A family from Leduc, Alta., is fighting for a treatment for their baby who has a rare genetic disorder.

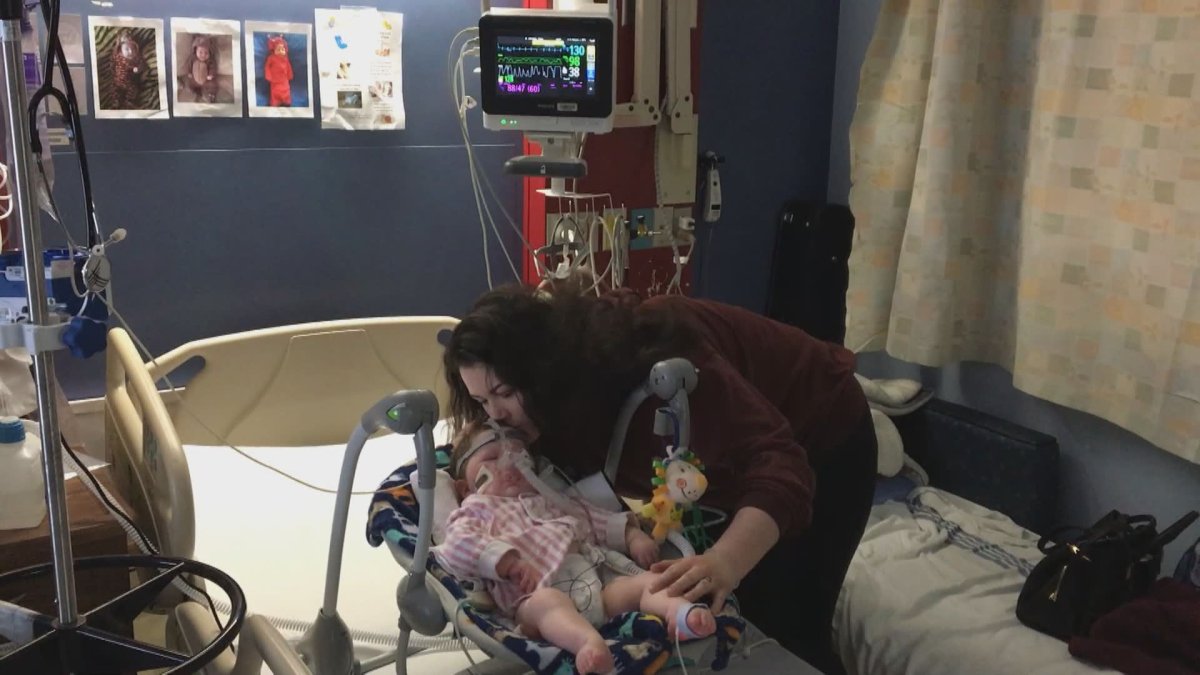
Alex Johnston and Ryan Fengstad are the parents of 10-month-old baby Reign, who was diagnosed with spinal muscular atrophy 1 (SMA1), which is also known as Werdnig Hoffman disease.
The disease essentially causes his muscles to waste away.
“As a parent, you see your child comes into this world, you have all these ideas and thoughts and dreams about what you’re going to do,” said Fengstad.
“That sort of came crashing down when we received the diagnosis.”
Most patients die before the age of two when their lungs fail.
“The amount of pain and stuff that he’s been through… he’s just done so well,” said Johnston.

Get weekly health news
Reign’s disease progression is currently being slowed by the drug Spinraza, which started to be covered by Alberta Health last year.
Now, the family has found a new treatment that could help Reign — but there’s one major issue. Not only is it currently not approved in Canada, but the treatment is also considered one of the most expensive therapies in the world.
- After controversial directive, Quebec now says anglophones have right to English health services
- Something’s fishy: 1 in 5 seafood products are mislabelled, study finds
- Why non-alcoholic beer is gaining steam at Oktoberfest: ‘Nobody will judge you’
- Recall expands for Nutrabolics vegan bars over undeclared milk
“We found out it was $2.8 million,” said Fendstad. “It was deemed the world’s most expensive therapy.”
Johnston and Fengstad hope to take Reign to the U.S. so he can be treated with the therapy, called Zolgensma.
It’s a one-time injection that replaces the patient’s missing gene so their body can make the protein it needs. It has to be administered before age two for it to be effective.
“But the whole point of making it possible is to save lives, isn’t it?”
“As we prepare to bring this treatment to Canadians, we are committed to working collaboratively with regulatory, pricing and reimbursement authorities in providing eligible patient access as quickly as possible.”
Novartis is also running a lottery-like program for patients outside of the U.S., giving away 100 doses for free for those in countries it isn’t yet approved in.
However, Reign’s parents are raising money to fund the treatment because they simply don’t have the time to wait for it to be available in Canada, and they can’t guarantee he will receive one of the free doses.
“The cost of it is overwhelming,” said Johnston. “But there’s hope.”
The family is hoping to raise money for the treatment through a GoFundMe page they launched.






Comments