Watch the video above: Light as a particle and a wave? How scientists were able to capture light behaving as both at the same time.

TORONTO — What is light? Most of us would think of light as a wave, right? Well, it is. But it’s also a particle.
Called the wave-particle duality, this scientific principle states that light exists as both a wave and particles, called photons.
READ MORE: WATCH: What does the sunset look like on Mars?
Scientists have photographed light in both states, separately, but never together. Until now.
When ultraviolet light hits a metal surface, it emits electrons, or subatomic particles with negative electricity. This is called a photoelectric effect.
So how’d this team of international scientists do it?

Get breaking National news
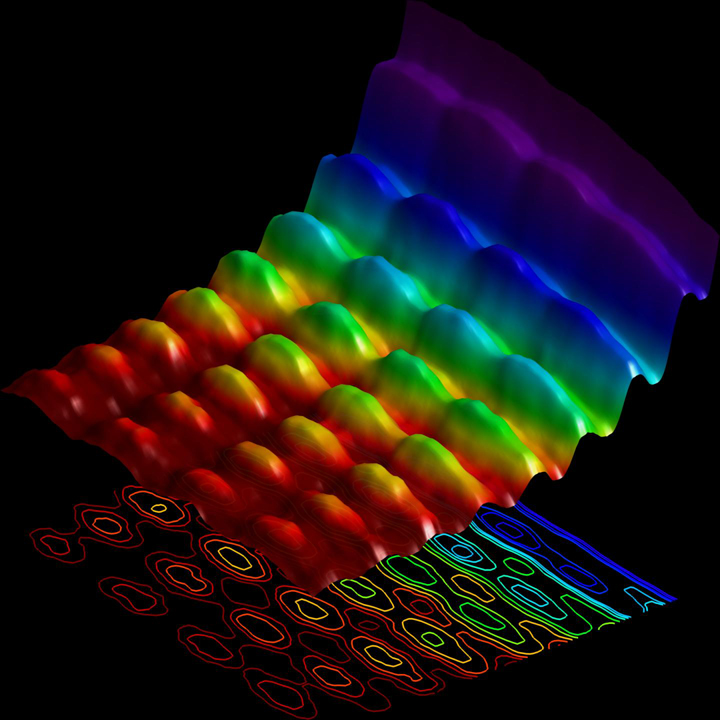
A pulse of laser light was fired at an extremely small metallic wire, called a nanowire. This added energy to the nanowire, causing it to vibrate. The light then travelled in two directions. When they met, it formed a new wave that seemed to be standing in place.
Then the researchers shot a stream of electrons close to the nanowire, imaging the standing wave of light. The electrons interacted with that standing light, either speeding up or slowing down. Using an ultrafast microscope, the scientists imaged the place where the change of speed occurred. That image is the wave of light.
Okay, so we have the wave, but where do the particles come in?
READ MORE: Climate scientist says rational threat assessment needed for nuclear power
Remember that change in speed? Well, as the electrons pass the wave they also hit the light’s particles, the photons. The change in speed is an exchange of energy packets between the electrons and the photons, which demonstrates that the light on the nanowire behaves as a particle.
So, aside from marking a science first, why does it matter?
The idea is that eventually this will help scientists develop more practical quantum computers which would be significantly faster than the computers we use today.
The paper was published in Nature Communications on March 2.







Comments
Want to discuss? Please read our Commenting Policy first.