Update: There is an update at the bottom of this story in regard to a “pre-print” study of four rapid tests that the federal government had distributed to provincial governments. Global News also has updated information in a story published on Feb. 29, 2024.

Health Canada ignored critical warnings about a rapid-test supplier before approving its COVID-19 kits for distribution nationwide, Global News has found.
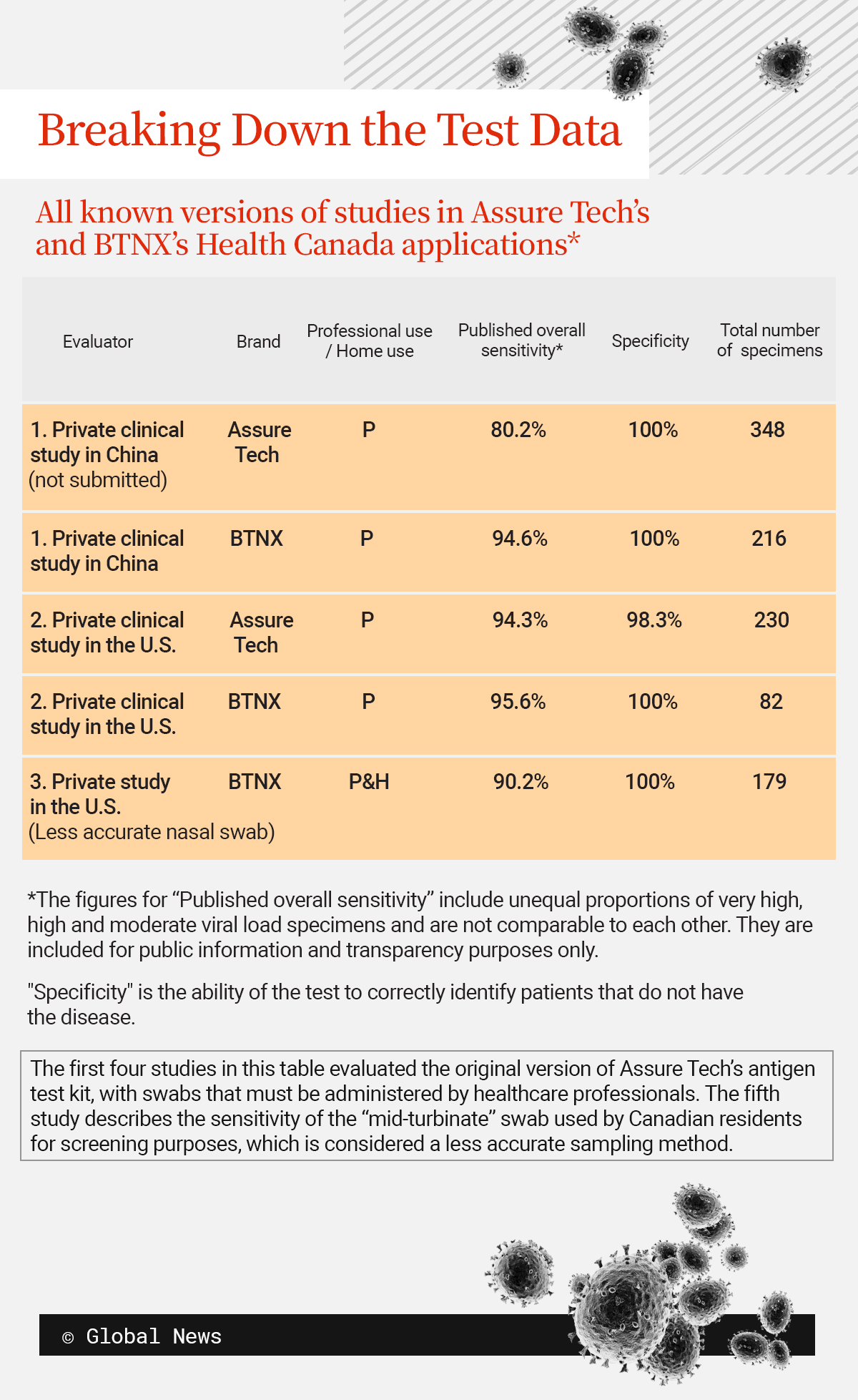
A year-long Global News investigation into federal procurement revealed that BTNX, a Toronto-area rapid-test supplier that buys the devices from China, deleted dozens of specimens, or samples, from a study it submitted to Health Canada in October 2020. Deleting the specimens increased the estimate of the rapid test’s ability to detect the virus.
In October 2020, BTNX submitted an application to Health Canada as it sought approval to sell its rapid test for COVID-19. One of the studies in the package was a study that suggested the test would detect 94.55 per cent of infections, among people who had symptoms for less than 14 days.
However, two months earlier, BTNX told authorities in Peru the same test detected 80.2 per cent of infections overall, according to records that government published online.
The lower detection rate of 80.2 per cent of infections is the estimate that the kit’s Chinese manufacturer, Assure Tech, gave to every business selling the test worldwide. It was based on a study of the test’s ability to detect the virus carried out at two health-care facilities near the company’s Hangzhou headquarters.
Though an assistant deputy minister at Health Canada had already flagged possible issues with another BTNX application, federal employees reviewing BTNX’s file did not challenge the company about this improvement or ask for an explanation, according to correspondence Global News obtained via freedom of information request.

BTNX admitted that it had deleted specimens. The company “worked with the data provided to us,” BTNX wrote to Global News in December 2023, making changes to “focus the data on the intended use of the product.”
In the wake of Global News’ reporting, BTNX added in a letter that the changes to the study were “based on the US FDA EUA template” and, in a press release, in “accordance with industry standards.”
The leap in the product’s accuracy was one of at least three red flags that Health Canada appears to have either missed or ignored as BTNX applied for a licence to sell the test kit in the fall of 2020. This included a warning from a senior Health Canada official about the veracity of the company’s public statements; data missing from a second study in BTNX’s application; and what researchers called elevated test results.
BTNX and Health Canada have stated that BTNX’s device works as promised, and regulators had all the information they required. Health Canada told Global News last month it will not re-examine BTNX’s licence to sell this test.
Dr. Stephen Ellis, member of Parliament for Cumberland-Colchester and the Conservative shadow minister for health, told Global News that his party will “hold the Liberals to account” and follow up on this “very serious concern regarding rapid test approvals.”
When Global News asked Mark Johnson, a Health Canada spokesman, what guidance applies to suppliers editing studies, he quoted the Food and Drugs Act.
“No person shall knowingly make a false or misleading statement,” he wrote.
Regardless, the federal government awarded BTNX a series of contracts in 2021 and 2022 that became Canada’s largest pandemic deal for medical supplies.
A wartime effort
When borders closed in the spring of 2020, with COVID-19 infections spreading, no vaccines, and entire nations in lockdown, workers at Health Canada and other federal offices faced a daunting task in finding the medical supplies Canada needed.
Government employees in Ottawa found themselves caught up in a global competition for rapid tests so fierce that a Health Canada official called it the “Hunger Games.”
Global News began looking into procurement while rapid test purchases were still underway. Federal ministries permitted some of the workers who evaluated rapid tests or procured them to participate in a rare group interview on the condition that Global News would not name employees or ask about contractors.
The interview took place in November 2022. Certain disparities in statements by BTNX executives a few weeks later led Global News to further investigate how and why BTNX received its contracts.
Six officials joined the interview, almost all of them at director level and above, in the interest of sharing their experiences during the public health emergency.
“The desperation can’t be captured in words,” recalled a senior official from the Public Health Agency of Canada.
All the world’s manufacturing plants were only able to meet a fraction of global needs. And Canadian officials could not match the purchasing power of their U.S. or European counterparts or the scale of their orders.
With few options, Canada turned inward. Prime Minister Justin Trudeau called on Canadian manufacturers to produce medical supplies, from face shields to ventilators.

Get weekly health news
Authorities preparing to launch mass screening pinned their hopes on a test manufactured by Spartan Bioscience, a company based in Ottawa. This DNA-based “lab-in-a-box” solution attempted to solve the problem of accuracy.
Health officials around the world had concerns about the reliability of the obvious alternative to lab tests, which were cheaply made rapid tests. In many markets these had never been regulated — and the World Health Organization would issue two warnings about them within the next six weeks.
Health Canada hired new employees, set up an emergency authorization process for health care equipment, and cut wait times on licences to sell medical supplies.
Over at Public Services and Procurement Canada, the invocation of the National Security Exemption empowered employees to skip most processes.
According to a CBC report published on April 12, 2020, exploring why Health Canada was lagging behind U.S. and European regulators, a BTNX executive said in an interview that the company was disappointed with the amount of time Health Canada was taking to approve its antibody test.
“We’ve seen the speed in which other, other health agencies around the world have been able to take these decisions,” BTNX’s CFO Mitch Pittaway said. “We think that Canada will hopefully be in a position to take a well-informed decision quite quickly.”
Pittaway was pitching an “antibody” test that detected the body’s reaction to the presence of COVID-19. Antigen tests, released soon afterward, detect the virus itself.
He noted that the company was selling the tests in the U.S. through an emergency program.
The next day, Pierre Sabourin, who was then an assistant deputy minister at Health Canada, flagged to Stephen Lucas, deputy minister, that he thought BTNX was misleading the public. Global News obtained correspondence between federal workers from a government website and freedom-of-information requests.
“Some manufacturers/importers claim falsely” that U.S. health authorities authorized their antibody tests for sale, Sabourin wrote. “For example, BTNX.”
At the time, the U.S. regulator was experimenting with pre-authorization sales, allowing rapid test suppliers to sell their products while they waited for officials to process their applications. BTNX was among that group.
BTNX’s application to Health Canada for authorization to sell the antibody test was “poor quality,” Sabourin added. The supplier had not submitted clinical data that backed up its claims, he wrote.
In response to Global News’ recent questions, BTNX wrote that it has always been transparent with regulators and the public.
On April 29, 2020, in a House of Commons debate, Prime Minister Trudeau called BTNX an “innovative Canadian company that has moved forward with a world-class product.”
Health Canada did not respond to Global News’ questions about whether the ministry informed the Prime Minister’s Office about Sabourin’s concerns.
In a statement issued after this article was published on Jan. 11, 2024, the Prime Minister’s Office wrote that BTNX’s antigen “rapid tests work well and have been successfully used by millions of Canadians” to fight COVID-19. A spokeswoman described Health Canada’s decision-making as “independent.”
A priority application
Six months later, Spartan’s device had not proven reliable enough, according to Health Canada’s evaluation. And any obstacles to BTNX’s applications had seemingly vanished.
David Boudreau, director general of Health Canada, emailed Sabourin and Manon Bombardier, assistant deputy minister, on Oct. 14, 2020, to announce that, after discussions, BTNX had applied for authorization to sell an antigen test for COVID-19.
Given the emphasis on finding antigen tests, he wrote, Health Canada would review the file “in priority, as soon as possible.”
It is unclear why Health Canada prioritized BTNX’s application.
Assure Tech, BTNX’s supplier in China, had already applied separately to Health Canada for authorization to sell exactly the same product.
Sabourin did not reply to Boudreau in writing.
(Sabourin declined Global News’ request for comment. He retired in December 2021.)
Federal employees set to work.
In Winnipeg, workers at the National Microbiology Lab evaluated sample tests BTNX provided.
In Ottawa, an employee with a PhD in biochemistry supervised the examination of BTNX’s and Assure Tech’s nearly identical application packages, which workers paired together, according to federal records.
One of the evaluations in BTNX’s package, but not in Assure Tech’s, was the edited version of the study of the test’s ability to detect COVID-19.
It was not immediately apparent that the two documents described the same study. Global News’ analysis showed that in addition to omitting data, BTNX had renumbered the locations at which the evaluation took place and the specimens’ identifying numbers.
A quick scan of Canadian distributors’ websites would have shown that another business was marketing the same test to customers overseas with the overall 80.2 per cent sensitivity estimate. Yet in BTNX’s application to Health Canada, this figure jumped to nearly 95 per cent.
“There was no switching of sites,” the company’s spokesman wrote. BTNX provided a “reliable testing tool for Canadians.”
One of the changes involved removing “retrospective positives.” This refers to specimens that have been frozen.
BTNX said Canadian and U.S. health regulators recommended using fresh samples.
In fact, Health Canada advised the company that it could use frozen specimens in its correspondence with BTNX. The U.S. FDA said the same.
Global News’ examination of the data also revealed that BTNX did not delete all of the frozen specimens, only those that tested positive for COVID-19. Those deletions improved the outcome for the nasal test by nearly 10 percentage points.
Examining clinical studies posted to Health Canada’s website, Global News found that other rapid test suppliers used retrospective samples and described the results from unusable specimens.
In response to questions about whether editing data was common among Canadian rapid test companies, Cenk Ozkan, vice president of Artron Laboratories, based in Burnaby, B.C., told Global News that deleting data would risk “loss of credibility and legal actions.”
Health Canada, for its part, has defended its decision to approve BTNX’s application. The approval relied on the full package, a spokesman wrote in December, including a second study of the test’s sensitivity that was carried out in the United States and managed by Assure Tech, BTNX’s supplier.
Global News discovered BTNX’s version of this second study was different from Assure Tech’s as well.
The version of this second study Assure Tech submitted to Health Canada cited 230 specimens. BTNX’s version cited just 82, suggesting that an unknown person omitted the majority of the specimens.
Assure Tech did not respond to Global News’ requests for comment. BTNX told Global News that Assure Tech provided only 82 specimens to BTNX.
“AssureTech did not advise BTNX that it had additional specimens from another site or that it was submitting a broader study to Health Canada,” the company’s spokesman wrote on Jan. 5. Health Canada’s approval of the test for consumers relied on a third evaluation in the package, he stated.
The deletions have raised questions among Canadian researchers about why the regulator approved BTNX’s application.
Trudo Lemmens, a professor of health law and policy at the University of Toronto, said, “The fact that Health Canada didn’t see it or ignored it” suggests “regulatory failure.”
Lemmens called on the federal government to launch an investigation that also allows for “scrutiny by independent researchers outside of government, outside of industry, to be able to see whether the data supports the claims.”
New questions about the application
In the November 2022 interview, a Health Canada official recalled federal workers’ sense of unity as he looked back on those terrible days.
He and his colleagues seemed confident that they had weeded out less reliable suppliers.
“We came to realize which companies were able to have quality applications” with “data that would support the claims,” he explained to Global News.
Since Global News’ report was published, Health Canada has argued that the National Microbiology Laboratory’s evaluation of BTNX’s kit supported BTNX’s stated sensitivity.
Global News obtained the NML’s results and the lab report, which called the device a “less-sensitive” test. The technicians attributed its lower outcomes to storage issues.
Unknown to Health Canada, researchers for the German, Spanish and British governments were separately reaching the same conclusion in the fall of 2020 and winter of 2021. (To learn more, you can read our Dec. 21 story online.)
Researchers Global News asked to review the data said the contrast between the governments’ results and the outcomes BTNX reported to Health Canada were eye-catching.
The ministry approved BTNX’s and Assure Tech’s applications on Feb. 19, 2021.
Over the next year and a half, the federal government bought 404 million test kits from BTNX for an estimated $2 billion. Global News’ reporting indicates that this was the biggest order any government placed with any supplier of Assure Tech’s device worldwide.
—
Update: On Jan. 7, 2024, a group of scientists primarily working for the Public Health Agency of Canada (PHAC) published a pre-print of a study of four rapid tests that the federal government had distributed to provincial governments.
A “pre-print” is a draft of a study posted before qualified scientists examine it, request revisions, and approve it for publication. A pre-print should not be used to guide medical practice. The study examined the tests’ ability to detect COVID-19, the Omicron variant, and several other variants.
According to the pre-print, the outcome that the team found for BTNX’s rapid test “corroborates manufacturer claims.” Press officers for PHAC did not inform Global News about the study before this article was published on Jan. 11, 2024. It is unclear which of BTNX’s tests were examined, when the study took place, or the extent of the agreement between PHAC’s study and those studies BTNX submitted to Health Canada.
Global News has asked PHAC for more information.











Comments
Want to discuss? Please read our Commenting Policy first.