A popular birth control implant that has long been advocated for use in Canada has now been approved by Health Canada, according to the agency.

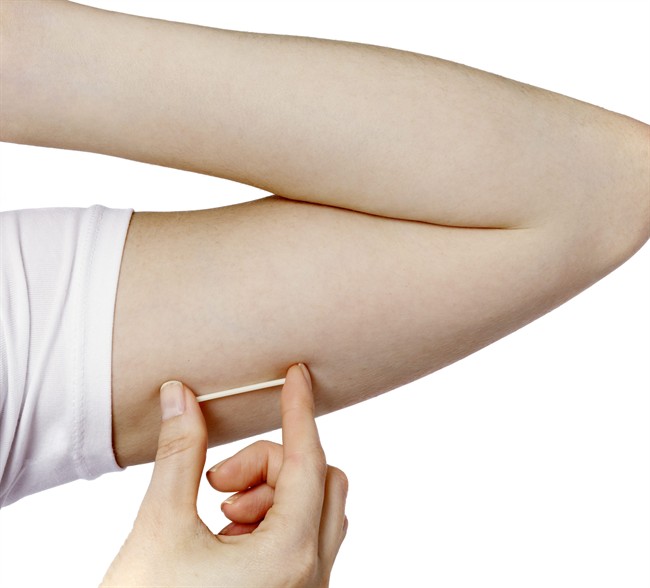
The use of Nexplanon, a three-year contraceptive implant that’s implanted into your upper arm, has long been unavailable in Canada as the research from its clinical trials did not yet meet Health Canada’s standards.
Mounting pressure to approve the contraceptive has been pushed by sexual health advocates for years, and has even resulted in an online petition directed at Prime Minister Justin Trudeau.
An emailed statement from a Health Canada spokesperson confirmed to Global News that the implant was approved on Monday, and has been added to the health agency’s drug product database which can be viewed online.
The implant’s popularity has been attributed to its accessibility as a contraceptive for women who are unable to take estrogen, as well the minimally-invasive process involved in both inserting and removing it.
Nexplanon and IUDs both fall under a category of birth control called long-acting reversible contraceptives (LARCs).

Get weekly health news
Robert Reid, an adjunct professor from the obstetrics and gynecology department at Queen’s University, told Global News in a previous interview that LARCs were great because “you put them in, you forget about them.”

“The problem is that putting in an IUD or an implant requires some skill and training… it’s a lot easier to reach in the cupboard and get the pills than try to make a referral that might take a month or two to get an IUD.”
Merck — the drug company that produces the contraceptive — has long billed the implant’s success in preventing pregnancies as over 99 per cent.
— With files from Meghan Collie








Comments