U.S. FDA
-
Sale of over-the-counter birth control pill backed by U.S. FDA panelFDA health advisers say a decades-old birth control pill should be sold without a prescription, paving the way for a likely U.S. approval of the 1st over-the-counter contraceptive.HealthMay 11, 2023

-
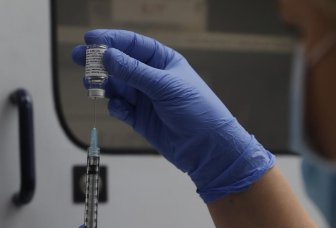
U.S. FDA advisors back bivalent COVID vaccine for initial series, boostersAdvisers to the U.S. Food and Drug Administration on Thursday unanimously voted in favor of targeting the same coronavirus strain for initial COVID-19 vaccine doses and boosters.HealthJan 26, 2023
-
-
Advertisement
-
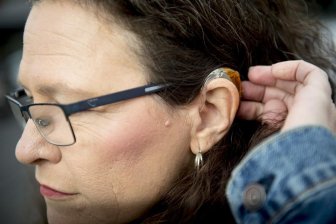
U.S. FDA allows over-the-counter hearing aids in ‘milestone’ regulationThe U.S. regulator said the new regulation cuts red tape by creating a new class of hearing aids that don't require a medical exam, a prescription and other specialty evaluations.HealthAug 16, 2022
![]()
-
-
Advertisement
-
Why Novavax vaccine may be an option for U.S. troops who refused other shotsThe Novavax vaccine may be an acceptable option for some of the 27,000 service members who have sought religious exemptions from the mandatory vaccine.HealthJun 9, 2022
![]()
-
U.S. FDA issues plan to ban menthol cigarettes, flavoured cigarsMenthol cigarettes, banned in many states including California and Massachusetts, account for more than a third of the industry's overall market share in the United States.HealthApr 28, 2022
![]()
-
-
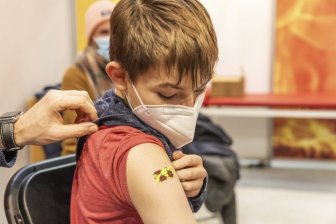
Pfizer seeks U.S. approval of COVID-19 booster for children aged 5-11Pfizer and BioNTech have filed for the clearance of a 10-microgram booster dose for children 5 to 11 years.HealthApr 26, 2022
![]()
-
-
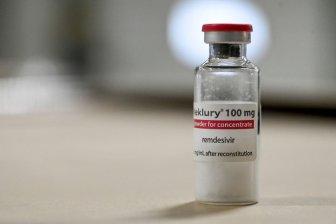
U.S. gives full approval to 1st COVID-19 drug for young childrenThe U.S. drug regulator on Monday granted the first full approval for treating COVID-19 in children aged 28 days and older to Gilead Sciences Inc's GILD.O drug remdesivir.HealthApr 25, 2022
![]()
-
New COVID-19 breath test approved in U.S. Here’s how it could be usefulA non-invasive devise using breath samples can test for COVID-19 in three minutes, but experts say there is a trade-off to the convenience and speed.HealthApr 16, 2022
![]()
-
-
Advertisement
-
U.S. FDA panel backs booster shot for Johnson & Johnson COVID-19 vaccineFDA's reviewers found no new safety concerns after a second dose of J&J against COVID-19, but they cautioned about the small number of patients studied and short follow-up period.HealthOct 15, 2021
![]()
Trending
-
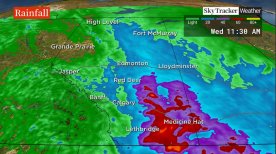
![]() A month’s worth of rainfall in 48 hours expected in some parts of Alberta30,119 Read
A month’s worth of rainfall in 48 hours expected in some parts of Alberta30,119 Read -
![]() Missing Australian, American surfers found dead in well with gunshot wounds14,603 Read
Missing Australian, American surfers found dead in well with gunshot wounds14,603 Read -
![]() Met Gala 2024: The most daring, dazzling and outrageous red carpet looks14,449 Read
Met Gala 2024: The most daring, dazzling and outrageous red carpet looks14,449 Read -
![]() Wegovy now in Canada: Who should (and shouldn’t) use the weight-loss drug12,968 Read
Wegovy now in Canada: Who should (and shouldn’t) use the weight-loss drug12,968 Read -
![]() Small grocers, co-ops receiving boost from Loblaw boycott: ‘A lot of anger’6,103 Read
Small grocers, co-ops receiving boost from Loblaw boycott: ‘A lot of anger’6,103 Read -
![]() Drake’s security guard shot outside of rapper’s Toronto home5,814 Read
Drake’s security guard shot outside of rapper’s Toronto home5,814 Read -