When researchers at the National Microbiology Laboratory in Winnipeg discovered that their mice just weren’t getting sick, even when exposed to Ebola – an incredibly deadly disease – they knew they might have something exceptional.

Although, they didn’t really mean for it to happen.
“It wasn’t really designed as a vaccine,” said Steve Jones, one of its inventors, but now this Canadian invention is being used by the World Health Organization to fight an outbreak of Ebola in the Democratic Republic of Congo.
“It was a lucky mistake. As are many of the things which happen in science.”
Lucky mistake
The National Microbiology Lab in Winnipeg is run by the Public Health Agency of Canada. And one of its divisions focuses on rare, but potentially deadly diseases. “Its role is to prepare Canada for the possibility of importing these diseases, but also to be ready and capable to respond to emerging or new viruses,” said Dr. Guillaume Poliquin, senior medical advisor of the lab.
Back in the early 2000s, one of those emerging viruses was Ebola. After Sept. 11, governments were newly concerned about potential terrorism threats, including bioterrorism, and they identified Ebola as a high priority, said Heinz Feldmann, another researcher who worked on PHAC’s project.
So, government money helped to fund studies into the disease.
WATCH: An Ebola outbreak continues to spread. As Abigail Bimman reports, the education campaigns are crucial — as several quarantined patients escaped from isolation.

One theory as to why Ebola was so deadly had to do with its glycoprotein: a mixture of protein and sugars that coats the outside of the virus. To test that theory, said Jones, some scientists had run experiments on the glycoprotein that showed that it caused tissue damage.
But Jones, who now directs the University of Saskatchewan’s School of Public Health, said the team wasn’t sure that the U.S. researchers had done the experiment properly, so they worked on a better method to test out the protein so that future experiments could achieve clearer results.
So they came up with a way to introduce the Ebola protein to a body without giving the animal Ebola.

Get weekly health news
Essentially, explained Poliquin, the researchers took a virus called VSV, or vesicular stomatitis virus, which shares some properties with Ebola.
“VSV is a benign animal virus that also coats its outside in a glycoprotein,” he said. So they removed the VSV glycoprotein coating and replaced it with Ebola’s.
Then they infected some mice with the new virus.
“Since they showed no ill effects, we decided to see whether they were protected from Ebola. And they were. 100 per cent. They didn’t even look slightly sick,” said Jones.
They tested it again with a single dose, and again over a shorter time period, and again even a half hour after the mice had already been exposed to Ebola. And the mice were generally fine.
“This was amazing. It’s not only that we have a vaccine for Ebola, we have a potential therapy that we can give if we know people have been exposed and they are significantly less likely to die,” he said.
“That was really quite a cool discovery. I’m not gonna lie. It was very cool.”
Waiting
The researchers did more studies in mice, then in 2005, they published the results of a study on macaques, which showed it effectively protected the primates against Ebola.
Then, not much happened. The vaccine was patented but spent years waiting to be tested on humans.
The researchers needed more money, said Feldmann, who now works in the United States as a senior investigator for NIAID. “Ebola was of little interest if not of no interest at that time, as many things are if they don’t have much impact or have impact that affects people far away from us. So there was no money.”
“It’s always easy to say after the fact that we should have done more early on, but it wasn’t a big thing. It just wasn’t a big thing.”
“Part of the issue is funding and part of the issue is also the nature of this vaccine,” said Poliquin. The unique vaccine was hard to manufacture, he said.
Jones found it “immensely frustrating” to watch outbreaks of Ebola and the related Marburg virus continue, knowing that there was something that could potentially help.
“Ute Stroher and I who worked together on the vaccine and also in Angola in the Marburg outbreak, knew we had a vaccine which could have potentially protected these people who we were seeing falling ill and dying in Angola,” he said.
“It was extremely upsetting. And we came back determined to do whatever we could to push this vaccine through.”
Convincing people to fund human trials – which are very expensive – was tough though.
Outbreak
That changed in 2014, as an Ebola outbreak spread across West Africa, eventually killing more than 11,000 people.
The vaccine was suddenly of interest again and went into human trials. “It was extremely exciting to see that happening and a bit nerve-racking because I believed in this 100 per cent and really wanted it to be successful,” said Jones.
“And it was also frustrating because I knew, if only the resources had been available earlier, we could have been deploying this vaccine as they are now in the DRC. And if that had happened, then thousands of people would have survived, I’m sure.”
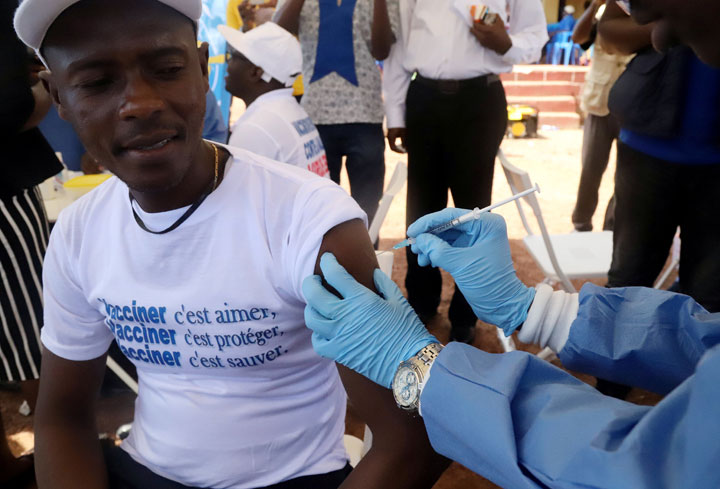
The trials were successful. And now, the World Health Organization has spent nearly a week vaccinating people in the DRC with the VSV vaccine, currently owned by Merck.
“To see it work, first in the clinical trials and then when they deployed it in the field, that was awesome,” said Jones.
WATCH: (Fri, Jul 31, 2015) The World Health Organization announced today that initial results of an Ebola vaccine trial in Africa show that it is 100 per cent effective after 10 days in preventing infection with the disease. Jen Tryon reports.
Feldmann said he was excited to see it too, but also thinks more work needs to be done. “Now questions come up. How durable is the immune protection and so on?”
It’s still experimental, first of all. There are also other vaccines which might work better in certain situations, he said, as well as a treatment (another Canadian invention) to help people who are already infected.
“It is a small contribution, but it is a contribution to global public health,” he said.
For people in the National Microbiology Laboratory, said Poliquin, “To be able to play that dual role of serving Canadians but also helping others throughout the world is a source of immense pride.”
Jones is elated. “To have potentially solved a problem like Ebola so the West African experience is never repeated? That’s awesome.”
“It doesn’t get any better than this.”










Comments
Want to discuss? Please read our Commenting Policy first.