Starting next year, Canadian women will be able to end their pregnancies with a pill.

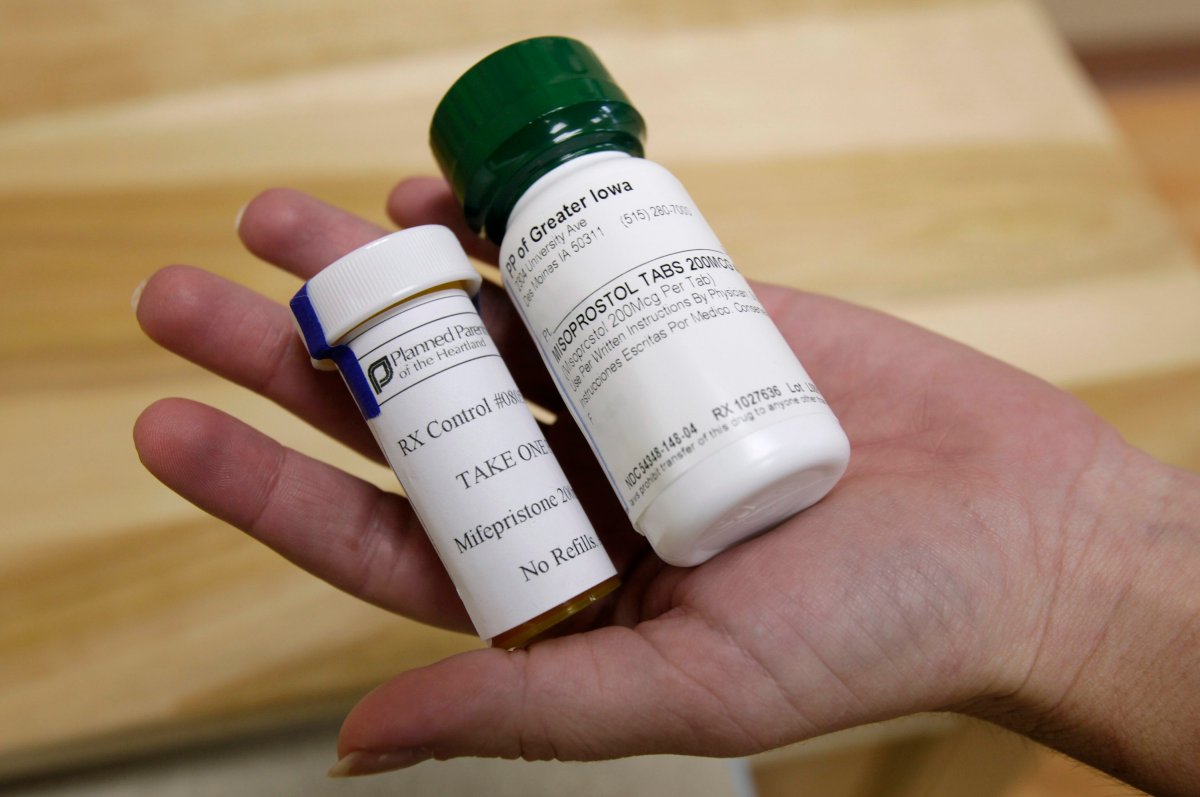
Earlier this year the federal government approved Mifegymiso, a combination of mifepristone and misoprostol better known as RU-486 designed to terminate a pregnancy within its first 49 days.
Women in France have had access to the so-called “abortion pill” for more than two decades. Ditto United Kingdom. American women have had it since 2000. And in many countries the pill is used as late as 70 days into a pregnancy, instead of the current 49-day limit here.
But in Canada it’s a new idea — one that could revolutionize access to reproductive health for women in remote and rural parts of the country.
In the wake of a fatal mass shooting at a Colorado clinic, as a new government takes the reins and as a new abortion pill rolls out next year, Global News took a closer look at who gets access to abortions — and who doesn’t.
ABORTIONS FOR SOME: Access still depends on who you are and where you live
Where can you get an abortion? It’s a secret
Provider patchwork: How abortion access varies across Canada
- ‘Alarming trend’ of more international students claiming asylum: minister
- TD Bank moves to seize home of Russian-Canadian jailed for smuggling tech to Kremlin
- After controversial directive, Quebec now says anglophones have right to English health services
- Why B.C. election could serve as a ‘trial run’ for next federal campaign
“Could” is the operative word. The company distributing Mifegymiso in Canada doesn’t know when in 2016 the rollout begins. And the restrictions placed on who can prescribe and dispense it limit its availability.
“Mifegymiso will be available sometime next spring, we are not certain yet of an exact date,” reads an emailed statement from Celopharma.
It can only be prescribed by a “trained and certified physician,” the email reads. “A patient can have a trained pharmacist fill a prescription, but, this prescription must be delivered directly to the physician and will never be in the hands of the patient.”

Get weekly health news
So this would expand abortion access if physicians who don’t provide surgical abortions get the training required to prescribe Mifegymiso, and the pharmacists they work with get the training needed to dispense it.
But if you’re in a remote or rural community, you don’t have a family doctor or your primary care provider doesn’t want to prescribe an abortifacent pill, you could be in the same boat, access-wise, you are now.
READ MORE: Where in Canada can you get an abortion? It’s secret
Canada’s pharmacists would like to see those restrictions loosened, at least enough to let pharmacists dispense the drug directly to patients.
“It has been our intent, because pharmacists are considered the drug experts, that pharmacists will play a greater role,” said Phil Emberley, director of pharmacy innovation for the Canadian Pharmacists Association.
The restrictions are in place for fear of side effects: Between two and five per cent of cases, Health Canada’s regulatory decision reads, the pill may not completely terminate a pregnancy, a woman might bleed more than normal or might need surgical intervention.
But Winnipeg-based doctor and abortion provider Suzanne Newman thinks expanding access to the pill “opens up all sorts of possibilities for northern women.” She envisions physicians prescribing it via telemedicine, and local pharmacists or nurses dispensing it and ensuring it’s taken as directed.
“That should increase access for women, rurally. Absolutely.”
Health Minister Jane Philpott wouldn’t comment on whether she’ll expand access to allow trained nurses and pharmacists to prescribe or dispense the medication.
“In July 2015, Health Canada authorized Mifegymiso for the termination of pregnancy up to 49 days following a thorough scientific review. The product is not yet available in Canada,” she’s quoted as saying in an emailed statement.
“The company will develop an education and registration program for prescribers and then advise Health Canada when the product is available for prescribing.”
PROVIDER PATCHWORK: How abortion access varies across Canada
How does the abortion pill work?
Mifegymiso is designed to initiate cervical dilation and uterine contractions, causing the uterus to expel the fetus and placenta.
Health Canada’s regulatory decision describes an average of 10.8 days of bleeding, including two days of “heavy bleeding,” and abdominal cramps that were bad enough to require pain medication in some women.
In addition to restricting its availability to specially trained physicians, Mifegymiso’s distributor agreed to “additional risk management measures,” including “a 24 hour patient support line, a patient consent form and distribution of Patient Medication Information to be provided to each patient.”







Comments